
Clinical trials can support better patient outcomes

Your patient’s health and
well-being is your priority.
Help them access potentially life-changing clinical trials.
Simple search & refer
It’s quick and easy to search Evripath for active clinical trials and refer your patients, just as you refer them to specialists.
Explore patient options
Evripath is designed for GPs. It makes exploring clinical trial treatment options for your patients an efficient and tailored process.
Contribute to medical research
Clinical trials help make global medical research breakthroughs happen. You play a critical role in referring patients who may benefit from participating in clinical trials.
Referral reimbursements
Evrima offers referral reimbursements for GPs who successfully refer patients who are the right-fit for clinical trial participation. Contact us to find out more.
They trust us
$1.4
Billion
The Australian Clinical Trials Sector Report (2021) released by MedTech and Pharma Growth Centre reported that $1.4 billion was spent on clinical trials in Australia in 2019. The sector now generates more export revenue than construction, intellectual property charges and government services.
1.9%
It’s estimated 487,901 participants registered for a clinical trial in Australia in 2020, or 1.9% of the population. Evrima is working to raise awareness of clinical trials and increase participation levels.

Evripath in action
Request a demoYour days at work are busy - and often long, too. Your patient’s health and wellbeing matters most, and you’d like to play a role in contributing to medical research if you can.
Learn how Evripath is empowering GPs to search and refer patients for vital clinical trials - all in a day's work at the medical practice.
Quick, easy and intuitive to use. Evripath is live now.
Get started with Evripath by following some simple and secure registration steps. Once you're logged in, you can search for active clinical trials that may be of benefit to your patients and refer them onto our experienced Evrima team for screening.
Register now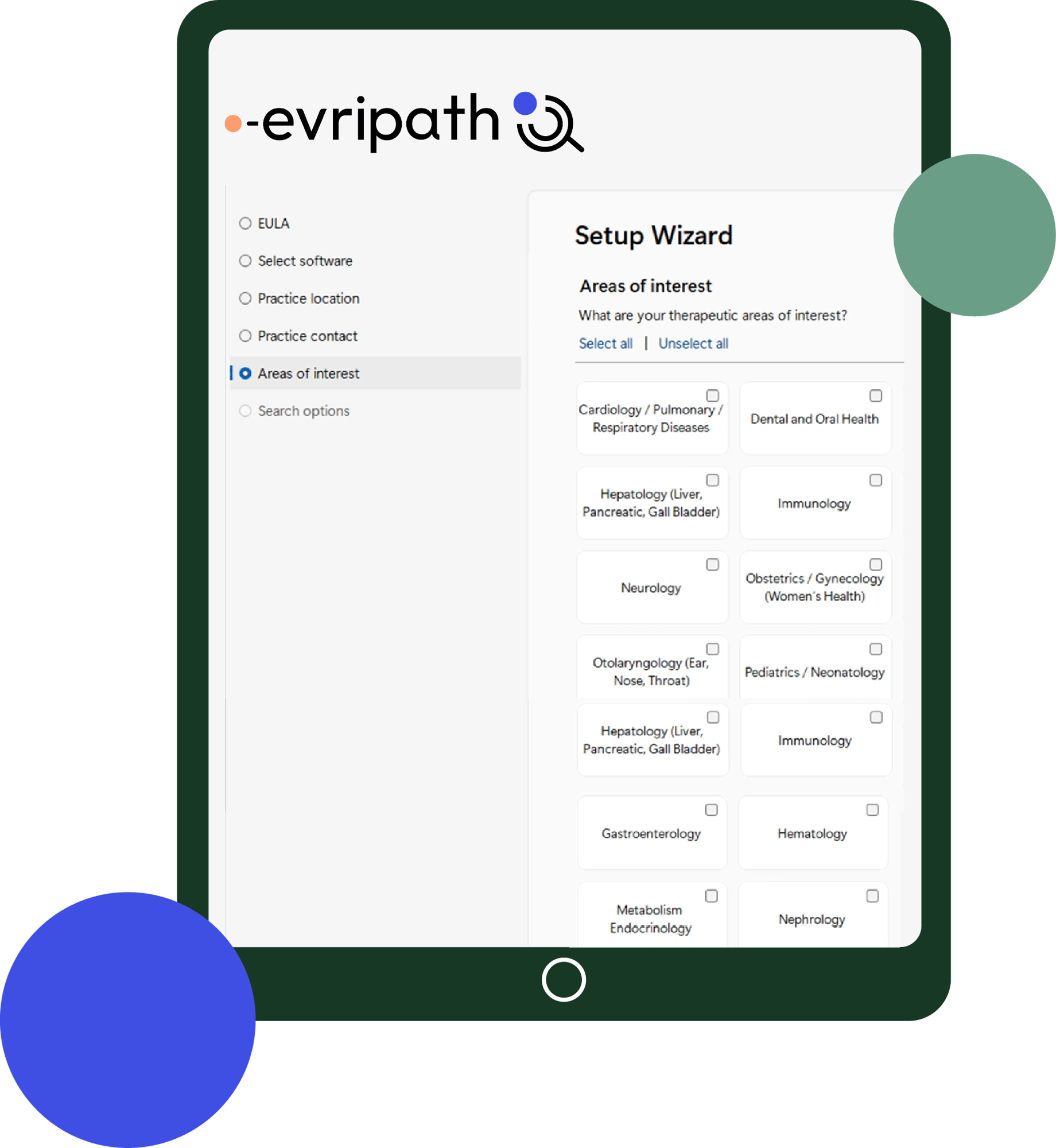
Make an impact with Evripath
Leading medical researchers
Evrima partners with leading researchers and clinicians around the world. You’ll be referring your patients to experienced, professional clinical trial teams.
Trusted clinical trials
All clinical trials that Evrima supports must go through an independent ethics committee review process and comply with Good Clinical Practice (GCP).
Access vast and varied trials
Use Evripath to search and refer your patients for clinical trials covering a vast range of therapeutic areas and chronic health conditions.
Screening support services
Evrima has an experienced team, including nurses, who can support your patients through the clinical trial screening process.
A better patient experience
Our team, along with our purpose-built Evrilink platform, ensures patients have visibility of their personal clinical trial registration progress, 24/7.
Research as a care option
You’ll be on the front foot with trial information to easily share with your patients. Have the option to discuss clinical research with your patients.
Advanced data protection
Evripath has advanced data security and privacy protections built-in. Evrima does not access your patient’s data, only the deidentified patient count is transmitted to us for software reporting purposes.
Refer your way
You can access Evripath yourself and discuss potential clinical trials with your patients, or Evrima staff can manage some of the patient communication for you. It’s your choice.
Evripath: FAQs and answers
01
What progress and patient updates will I receive from the clinical trial?
-
Trial Sponsors can provide a recruitment and progress update to GPs and you can also view your patient’s clinical trial journey via the secure Evripath platform.
02
What clinical trials are available for my patients?
+
Evrima works with leading researchers and clinicians to help make medical research breakthroughs happen and deliver better health outcomes. All clinical trials must go through an independent ethics committee review process and comply with Good Clinical Practice (GCP). Our trials span across a vast range of therapeutic areas and chronic conditions. You can view our active trials here.
03
How much time does referring a patient for a clinical trial take?
+
The power, choice and control is in your hands. Your medical practice can tailor the Evripath platform to suit how your practice likes to work, and we’re here to support you.
GPs or practice administrative teams can easily access trial information and discuss the trial options with eligible patients. Alternatively, your practice can engage Evrima staff to manage patient communication from within the practice.
GP discussions can occur in consultation or as part of standard recall.
Each option requires a varying level of involvement from the doctor and the practice team. The Evrima team is able to discuss the options with you and confirm your preferred approach to managing patient referrals and communication.
04
How do I install the Evripath GP search and refer platform?
+
It’s quick and easy. Contact us and you’ll receive simple instructions on how to download the Evripath search and refer platform. The Evrima team is here to answer your questions and help you get started.
05
What are the software requirements for the Evripath GP clinical trial search platform?
+
|
Software Name |
Version |
|
Operating System |
Windows 7 or newer |
|
Best Practice |
1.9.1.863 version or newer |
|
Medical Director |
4.1 version or newer |
06
Does Evrima access my patient data?
+
Evrima does not access your patient’s data. Only the deidentified patient count is transmitted to Evrima for software reporting purposes. As the GP, you remain in control of the patient data and how, if at all, the patient is contacted. The platform has advanced data protection built-in. We are GCP compliant, have a robust Privacy Policy and Terms of Service.
Latest news
No Results Found
Featured resources


